|
Results of murine NK cell studies are not always applicable to the human NK cell system because of the lack of a murine homologue of CD56 and because of differences in the inhibitory and activatory receptors. However, understanding the development and function of human NK cells is largely based on in vitro analyses, and models to study human NK cells in vivo are lacking. 10, 11 Other important regulatory cytokines are IL-12 and IL-18, which enhance cytotoxicity and trigger cytokine release by mature NK cells. FL and SCF play important roles in the early differentiation steps of NK cells and in their subsequent expansion and functional maturation as revealed in mice rendered deficient in FL or carrying mutations in the c-kit receptor. 6 Targeted disruption of the IL-15 or IL-15 receptor genes in mice 7, 8 and spontaneously occurring mutations in the signaling components of the receptor in humans 9 cause blocks in early NK cell development. 3-5 IL-15 is a key cytokine in NK cell development. These precursors can efficiently generate mature NK cells in vitro in the presence of interleukin-15 (IL-15) and early-acting cytokines such as flt3 ligand (FL) or stem cell factor (SCF), which increase the frequency of NK cell precursors responding to IL-15. NK cell precursors have been identified within the CD34 + hematopoietic cell population in adult bone marrow (BM) and umbilical cord blood (CB). 2 In contrast to T lymphocytes, NK cells are able to kill cancer and virus-infected target cells without the need for prior antigen stimulation. 1 They are important effectors of the innate immune system and contribute to the first line of defense against infections and malignancy. Human natural killer (NK) cells comprise approximately 10% of peripheral blood (PB) lymphocytes and are characterized phenotypically by the expression of CD56 and the lack of CD3 cell surface antigens. These studies establish conditions to explore human NK cell development and function in vivo in the NOD/SCID mouse model. Our results demonstrate that limitations in human NK cell development in the murine microenvironment can be overcome by treatment with NK cell growth–promoting human cytokines, resulting in the maturation of IFN-γ–producing cytotoxic NK cells. In the absence of growth factor treatment, CD56 + cells were present only at background levels, but CD34 +CD7 + and CD34 -CD7 + lymphoid precursors with NK cell differentiation potential were detected in BM and spleen of chimeric NOD/SCID mice for up to 5 months after transplantation. Human NK cells of NOD/SCID mouse origin closely resembled NK cells from human PB with respect to phenotypic characteristics, interferon (IFN)–γ production, and cytotoxicity against HLA class 1–deficient K562 targets in vitro and antitumor activity against K562 erythroleukemia in vivo. After 1-week-long in vivo treatment with various combinations of interleukin (IL)–15, flt3 ligand, stem cell factor, IL-2, IL-12, and megakaryocyte growth and differentiation factor, CD56 +CD3 - cells were detected in bone marrow (BM), spleen, and peripheral blood (PB), comprising 5% to 15% of human CD45 + cells. Here we describe conditions leading to the development of human NK cells in NOD/SCID mice receiving grafts of hematopoietic CD34 + precursor cells from cord blood. 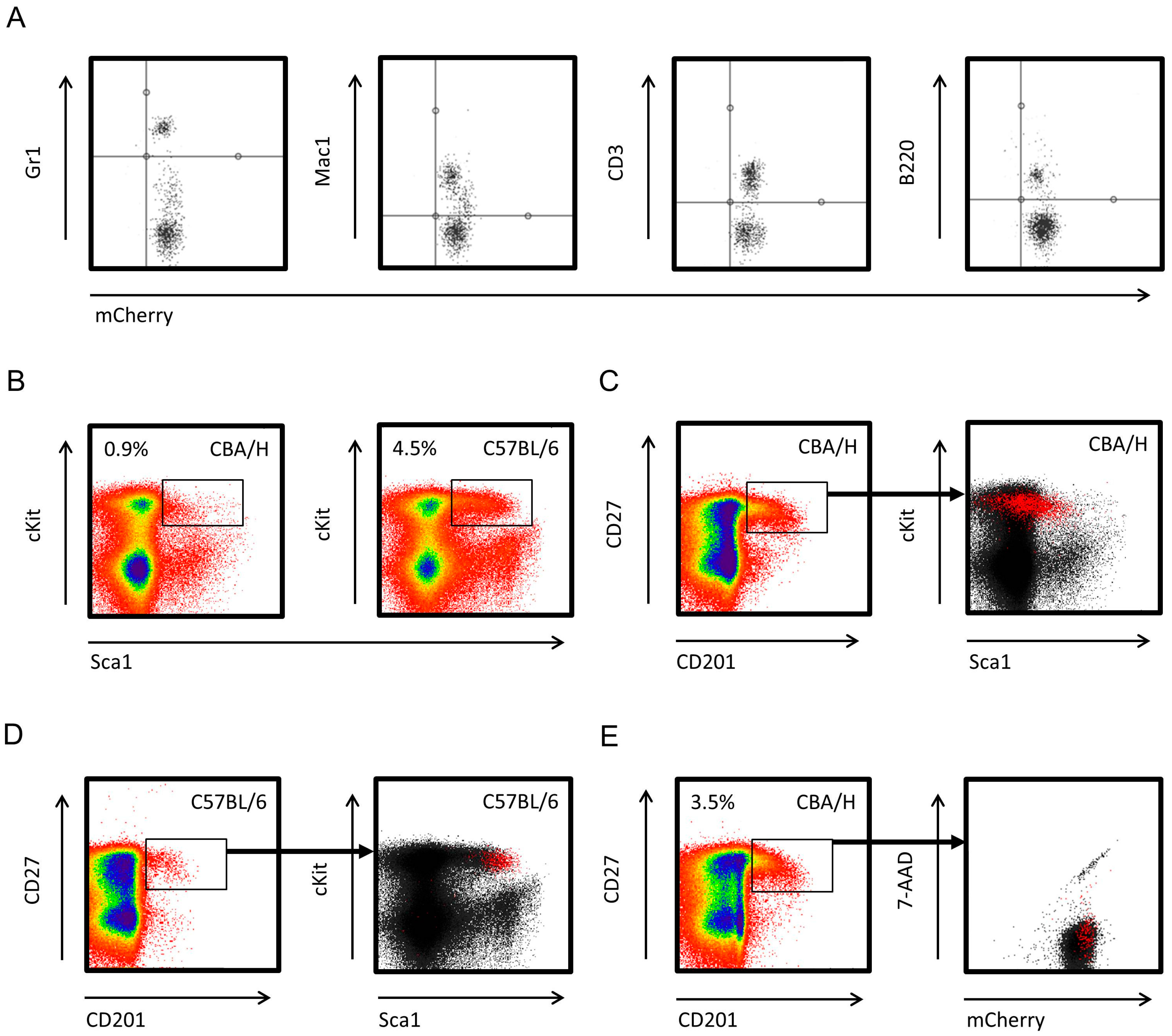
Definition of the cytokine environment, which regulates the maturation of human natural killer (NK) cells, has been largely based on in vitro assays because of the lack of suitable animal models.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
